How Many Valence Electrons Are in P3-
USE THE PERIODIC TABLE. Step 1 of 3.

P And P 3 Electron Configuration Phosphorous And Phosphide Ion Youtube
Answer to Solved How many valence electrons are in P3.

. Therefore Its valence electrons should be 5. Can anyone assist me with this question. Phosphorous is in Group 15 also called 5A and therefore Phosphorous has five valence electrons note that this method doesnt work for the Transition Metals.
Since the last shell of a phosphorus ion has eight electrons the valence electrons of phosphorus ionP 3- are eight. The phosphide ion P3- has a total of 8 valence electrons. Compound formation of phosphorusP PhosphorusP participates in the formation of bonds through its valence electrons.
Find the element that has 3 Valence Electrons and 2 energy levels. How many valence electrons does Phosphorus have. Phosphorus ionP 3- electron configuration After arranging the electrons it is seen that the last shell of the phosphorus atom has five electrons.
We review their content and use your feedback to. Experts are tested by Chegg as specialists in their subject area. How many valence electrons does the P3 ion have.
Valence electrons are those in the outermost shell of an atom. Lets check using the electron configuration. P3- ion has 15 protons as the atomic number of phosphorus is 15 and 18 electrons 15 electrons from phosphorus and three from the negative charge.
1s2 2s2 2p6 3s2 3p3 15 electrons atomic number of P The outermost orbitals 3s2 3p3 contains 5 electrons. Thus phosphorus has five valence electrons. Thus valence electrons for P is 5.
Report an issue. There are a total of five electrons present in the valence shell of phosphorus 3s²3p³. That gives a total of 5 electrons so neutral phosphorus atoms have 5 valence electrons.
Who are the experts. According to the periodic table above phosphorus belongs to Group 5A. The highest-numbered shell is the third shell which has 2 electrons in the 3s subshell and 3 electrons in the 3p subshell.
Ions are formed when atoms gain or lose valence electrons. How many electrons does phosphorus have in its valence shell. How many valence electrons does P 3-have.
Atoms will tend to gain or lose electrons so that they acquire a noble gas electron configuration normally containing 8.
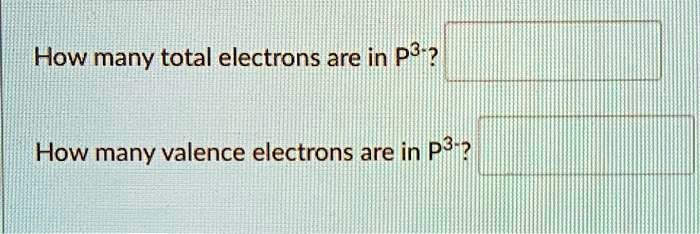
Solved How Many Total Electrons Are In P3 How Many Valence Electrons Are In P3
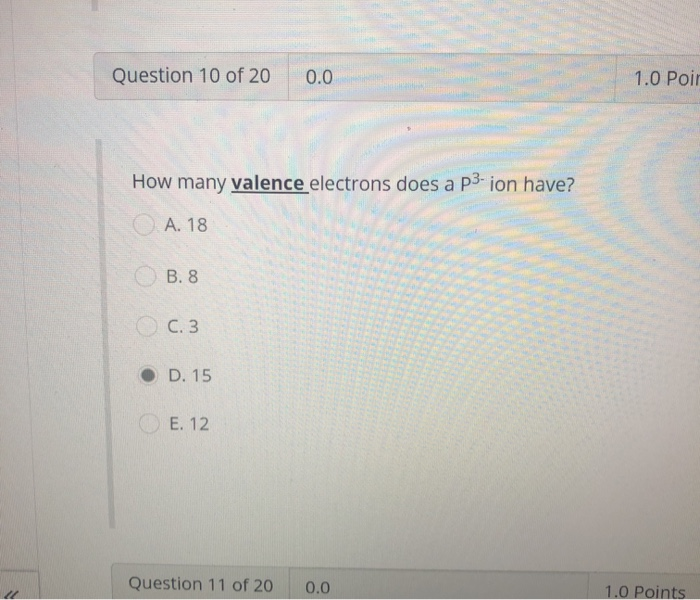
Solved Question 10 Of 20 0 0 1 0 Poir How Many Valence Chegg Com

How To Draw The Lewis Dot Structure For P 3 Phosphide Ion Youtube

Solved Question 14 Identify The Number Of Valence Electrons Chegg Com
No comments for "How Many Valence Electrons Are in P3-"
Post a Comment